- Blog
- Jailbreak free vip server links
- Afterglow ps3 controller pc turbo setup
- Battleship game online free
- Librecad change line width of selected lines
- Sakura dungeon uncensor steam patch
- Minecraft bedwars map download 1-12
- 2021 irs income tax brackets
- Funny zoom virtual background
- Sports head basketball 66 games
- Change text color in final cut pro
- Wicked whims download sims 4 1-44-77
- Google play ftp server
- Printable free daily preschool schedule template
- Bubble letters cute fonts to draw alphabet
- Mri safe dorsal column stimulator
- Eagle eye lights
- Computer notes hindi pdf
- Sony vegas pro 13 patch free download
- Mmpi 2 rf test questions pdf
- Create a pdf signature stamp
- Text art copy and paste generator
- Arc xt pro software download free
- Schivelbusch railway journey summary
- Daily schedule at kid
- Helvetica font free download for windows 10
- O2jam online
- Sketchpad online browser extension
- Ccleaner pro full crack android
- Setpoint for logitech mouse m705
- Review alien skin snap art
- Zenonia 5 mod apk offline
- 3d custom girl evolution mod download
- Calculate linear regression equation
- Gamemode 1 hacks minecraft bedrock
- Massage map adult erotic
- Periodic table valence electrons groups
- Free music download youtube downloader mp3
- Audacity record two sources at once
- Adding sticky notes to desktop mac
- Fl studio mobile 3 plugins free download
- Charles barkley-s wife brett favre daughter instagram
- List of sight words for kindergarteners
- Images of minion butts
- Bandwidth vs speed test
- Ds 160 form filling for b2
- Luminar 2018 lut download
Atoms become larger going down a group and going from right to left across a period. The relative sizes of the atoms show several trends with regard to the structure of the periodic table. Atomic radii of the representative elements measured in picometers. We know that as we scan down a group, the principal quantum number, n, increases by one for each element.\) Trends on the Periodic Table. General trends noted are increasing circle size moving from top to bottom in a group, with a general tendency toward increasing atomic radii toward the lower left corner of the periodic table. No spheres are provided for the noble or inert gas, group 18 elements. Beneath the molecule is the label, “I radius equals 266 p m divided by 2 equals 133 p m.” In figure b, a periodic table layout is used to compare relative sizes of atoms using green spheres.
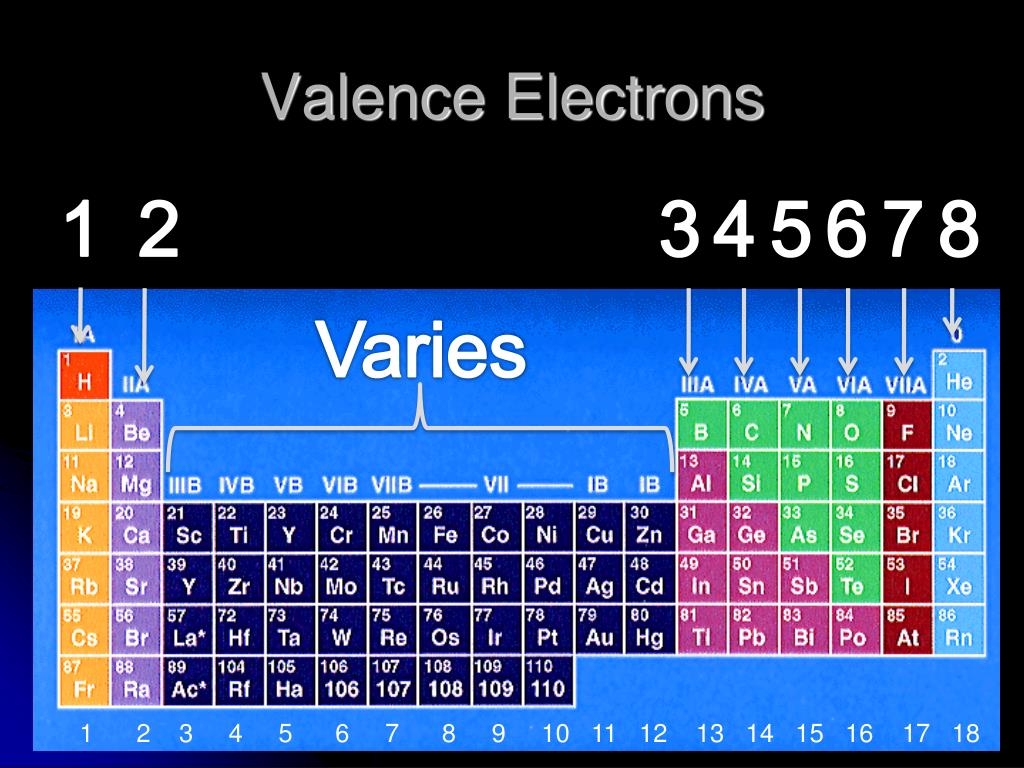
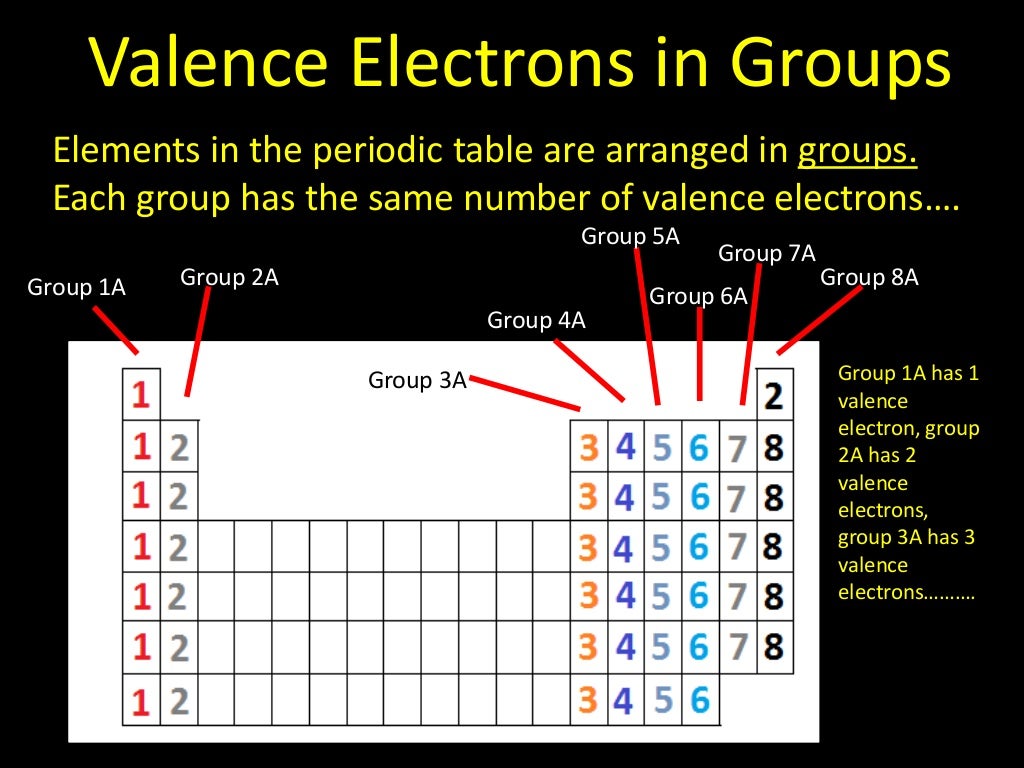
The distance between the radii is 266 p m. Using the ionization energies given below, determine the number of valence electrons this element has. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. Some groups on the periodic table have a name, since the elements in a single group have similar properties. The distance between the radii is 228 p m. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. Thus, Na in Group IA has 1 valence electron, whereas C, in Group IVA has 4 valence electrons. Conveniently, the number of valence electrons for the A Group elements is equal to the group number.
The distance between the radii is 198 p m. For the A Group elements (IA-VIIIA), the the valence electrons are those electrons in the s and p subshells of the highest energy shell. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. Using the variable n to represent the number of the valence electron shell, write the valence shell electron configuration for each group. Two spheres are pushed very tightly together. The shape of the periodic table reflects the order in which electron shells and subshells fill with electrons. The first model, in light green, is used to find the F atom radius. You could count how many groups to the right copper is to find how many.
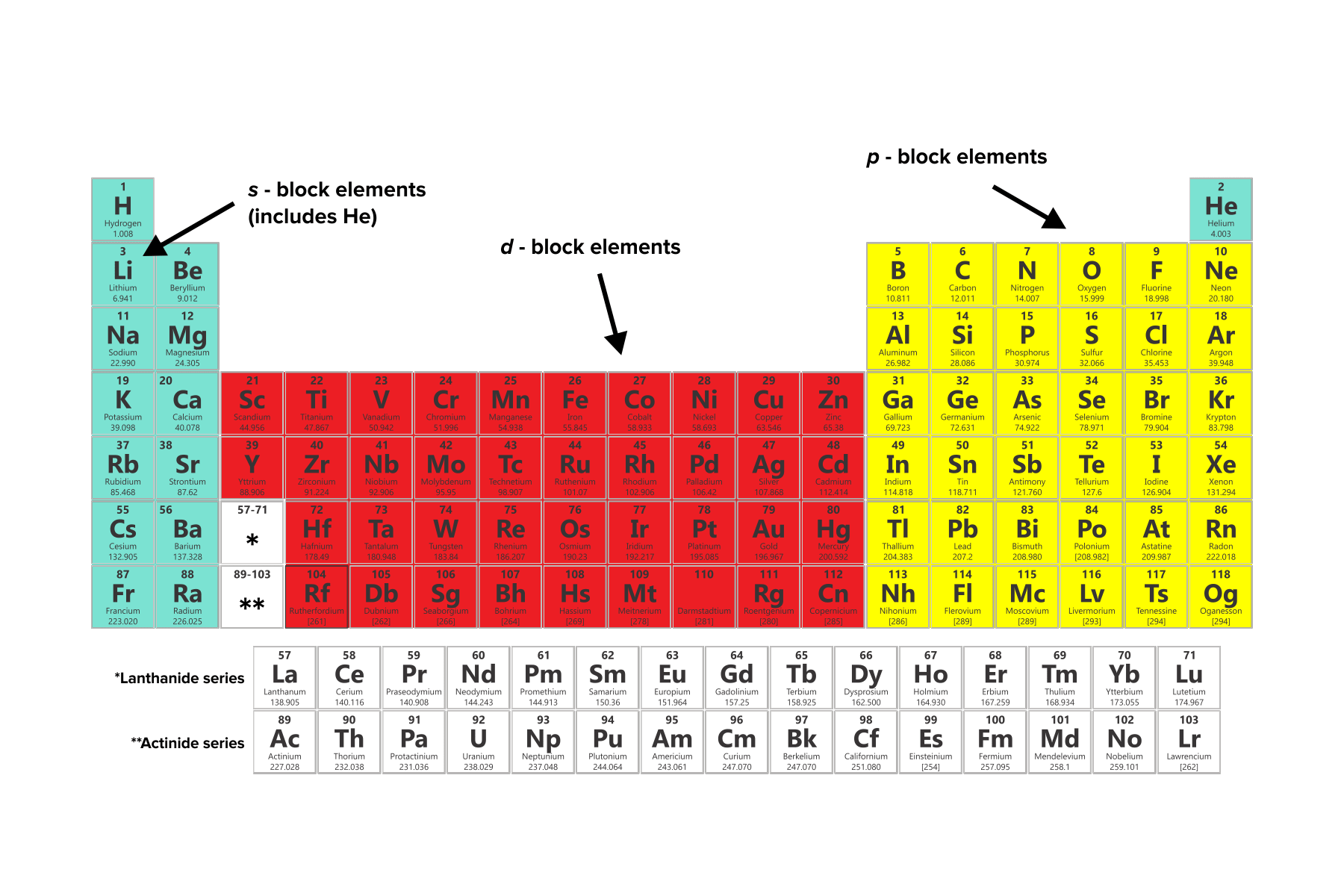
a transition metal in the fourth period like copper, Cu, this would mean a 4s and 3d orbital. Valence electrons are the electrons in the outermost shell, or energy level, of an atom.
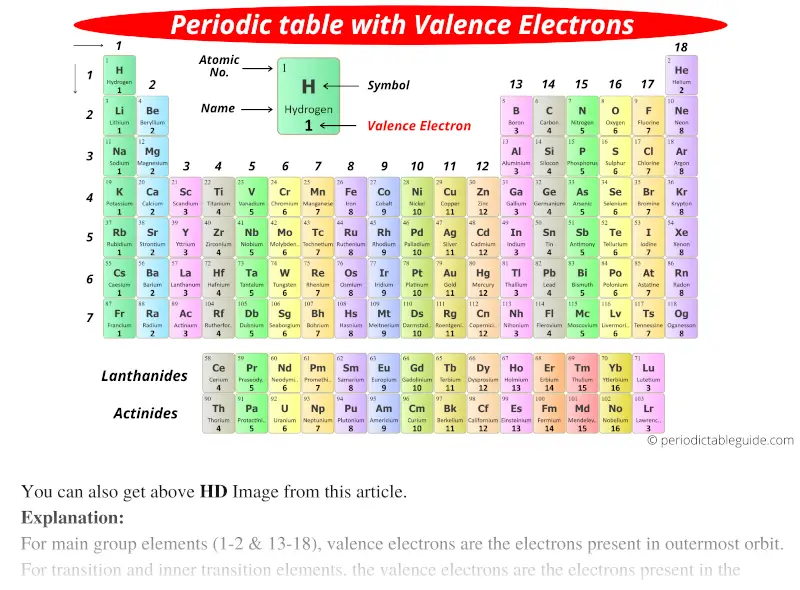
When we talk about a group, were just talking about a. The periodic table, electron shells, and orbitals. And in particular, were going to focus on groups of the periodic table of elements. We can use this method to predict the charges of ions in ionic compounds. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. For example, fluorine has seven valence electrons, so it is most likely to gain one electron to form an ion with a 1- charge. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. \): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
